 This relationship can be used to convert between temperatures in Fahrenheit and Celsius (see Figure 11.2). Since 100 Celsius degrees span the same range as 180 Fahrenheit degrees, one degree on the Celsius scale is 1.8 times larger than one degree on the Fahrenheit scale (because 180 100 = 9 5 = 1.8 180 100 = 9 5 = 1.8 ). 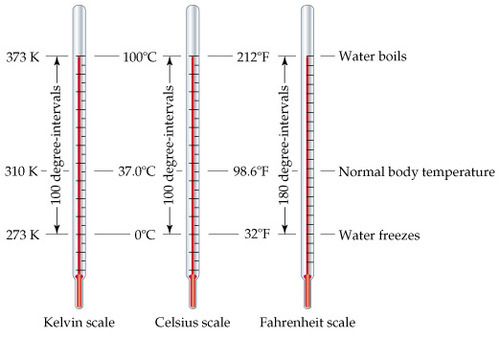
Therefore, a temperature difference of one degree Celsius is greater than a temperature difference of one degree Fahrenheit. Note that the difference in degrees between the freezing and boiling points is greater for the Fahrenheit scale than for the Celsius scale. The unit of temperature on this scale is the degree Fahrenheit ( ☏ ( ☏ ). On the Fahrenheit scale, the freezing point of water is at 32 ☏ ☏, and the boiling point is at 212 ☏ ☏. The boiling point of water is 100 ☌ ☌ for the Celsius scale, and its unit is the degree Celsius ( ☌ ( ☌ ). For example, the Celsius scale uses the freezing point of water as its reference point all measurements are either lower than the freezing point of water by a given number of degrees (and have a negative sign), or higher than the freezing point of water by a given number of degrees (and have a positive sign). Both the Fahrenheit scale and Celsius scale are relative temperature scales, meaning that they are made around a reference point. The three most commonly used temperature scales are the Fahrenheit, Celsius, and Kelvin scales. To measure temperature, some scale must be used as a standard of measurement. Take an eraser and rub it vigorously against any surface. You can show that temperature is related to the kinetic energy of molecules by a simple demonstration. Thermal energy is one of the subcategories of internal energy, as is chemical energy. The total internal energy of a system is the sum of the kinetic and potential energies of its atoms and molecules. Faster moving molecules have greater kinetic energies, and so the substance has greater thermal energy, and thus a higher temperature. Without going into mathematical detail, we can say that thermal energy-the energy associated with heat-is the average kinetic energy of the particles (molecules or atoms) in a substance. Recall that kinetic energy is the energy of motion, and that it increases in proportion to velocity squared. Since heat, like work, transfers energy, it has the SI unit of joule (J).Ītoms and molecules are constantly in motion, bouncing off one another in random directions. This is because we are sensitive to the flow of energy by heat, rather than the temperature. For example, we may say that the heat was unbearable, when we actually mean that the temperature was high. Temperature is literally defined as what we measure on a thermometer. Temperature is defined in terms of the instrument we use to tell us how hot or cold an object is, based on a mechanism and scale invented by people. Heat is the transfer of energy due to a temperature difference. It is tempting to say that temperature measures heat, but this is not strictly true. 
What is temperature? It’s one of those concepts so ingrained in our everyday lives that, although we know what it means intuitively, it can be hard to define.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
